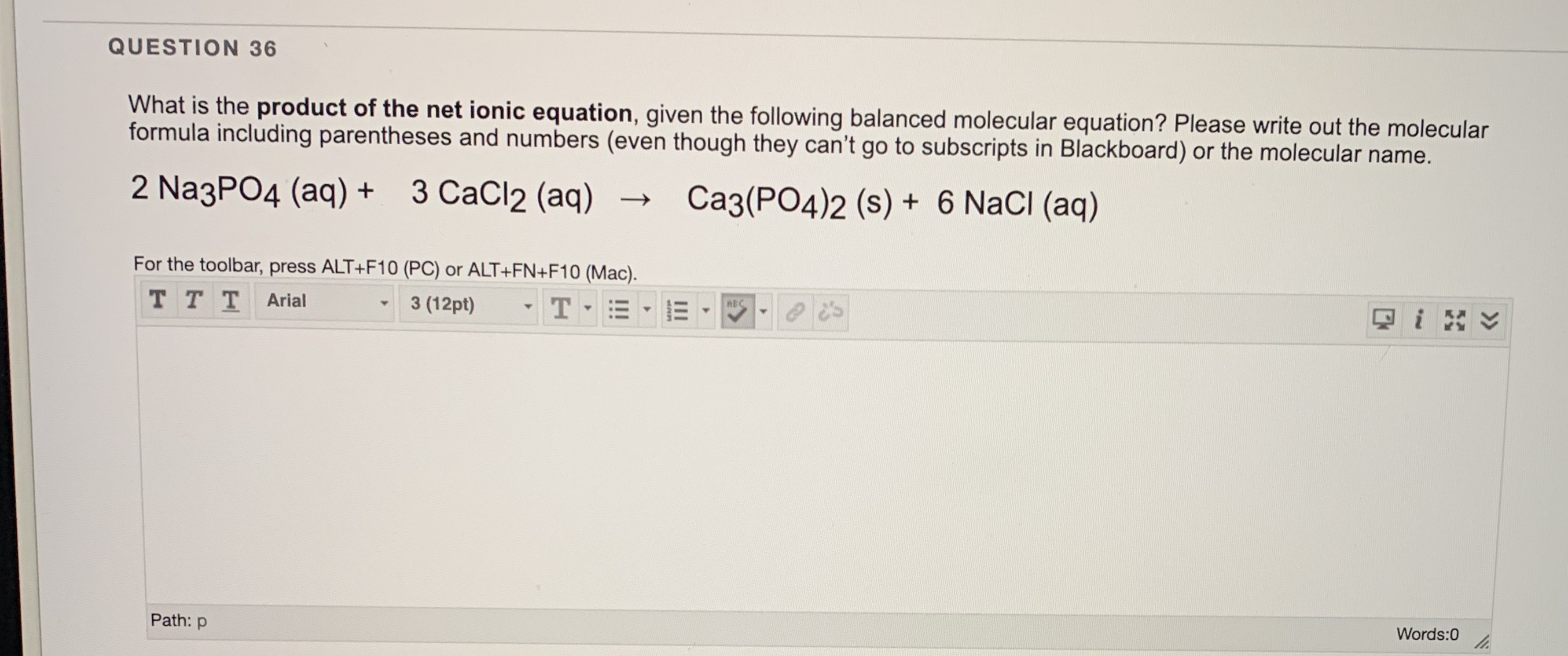
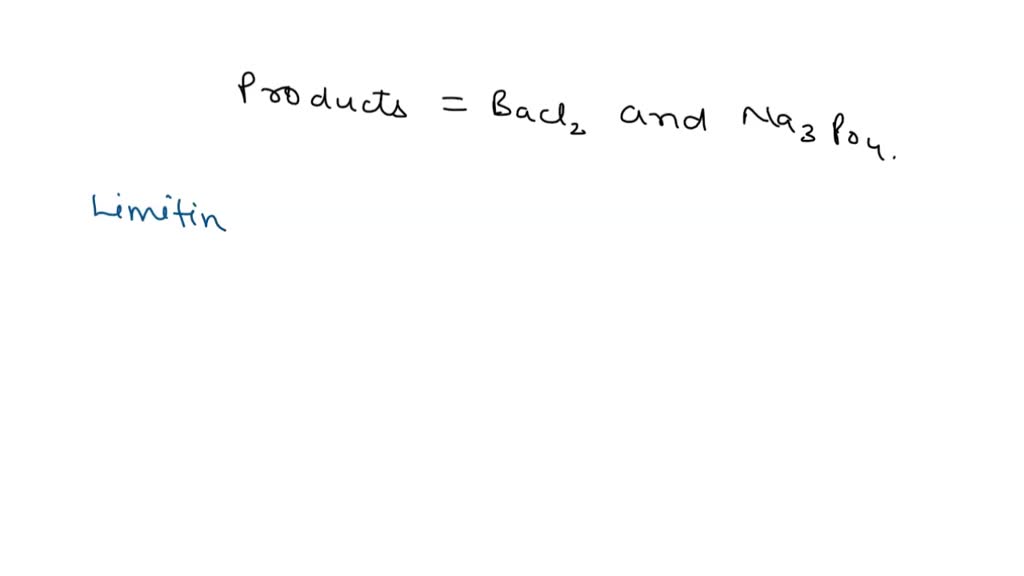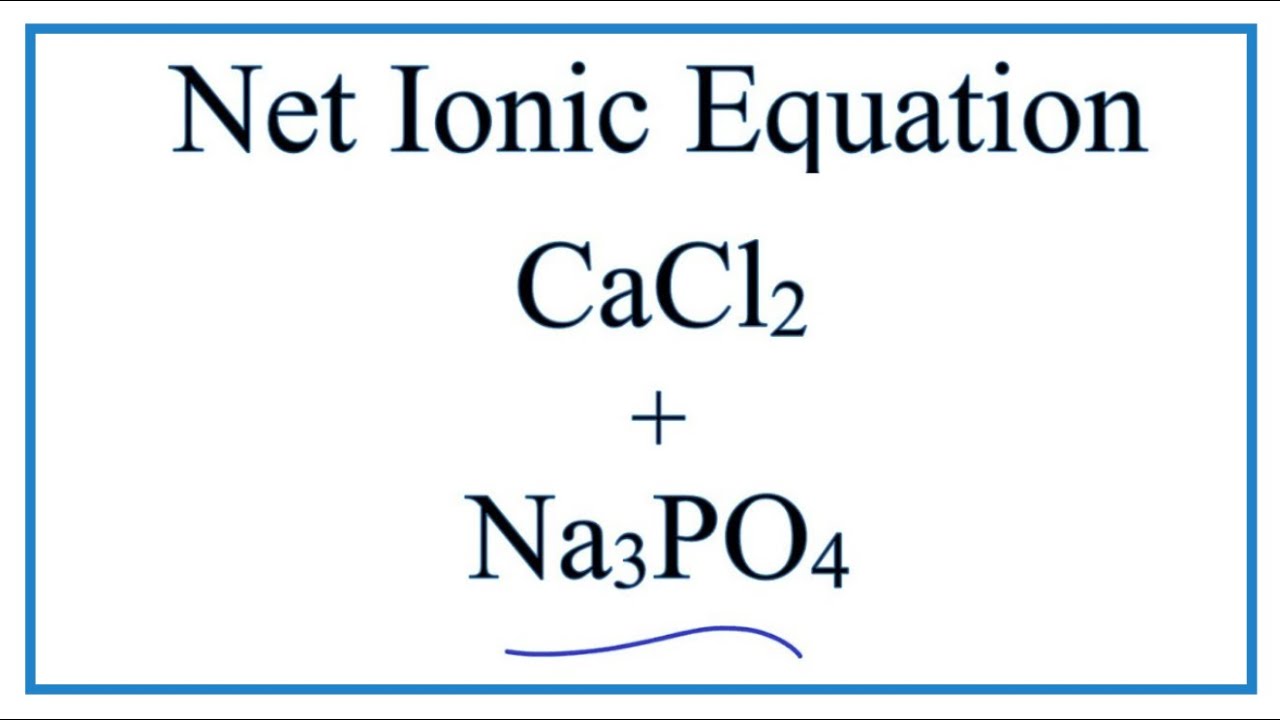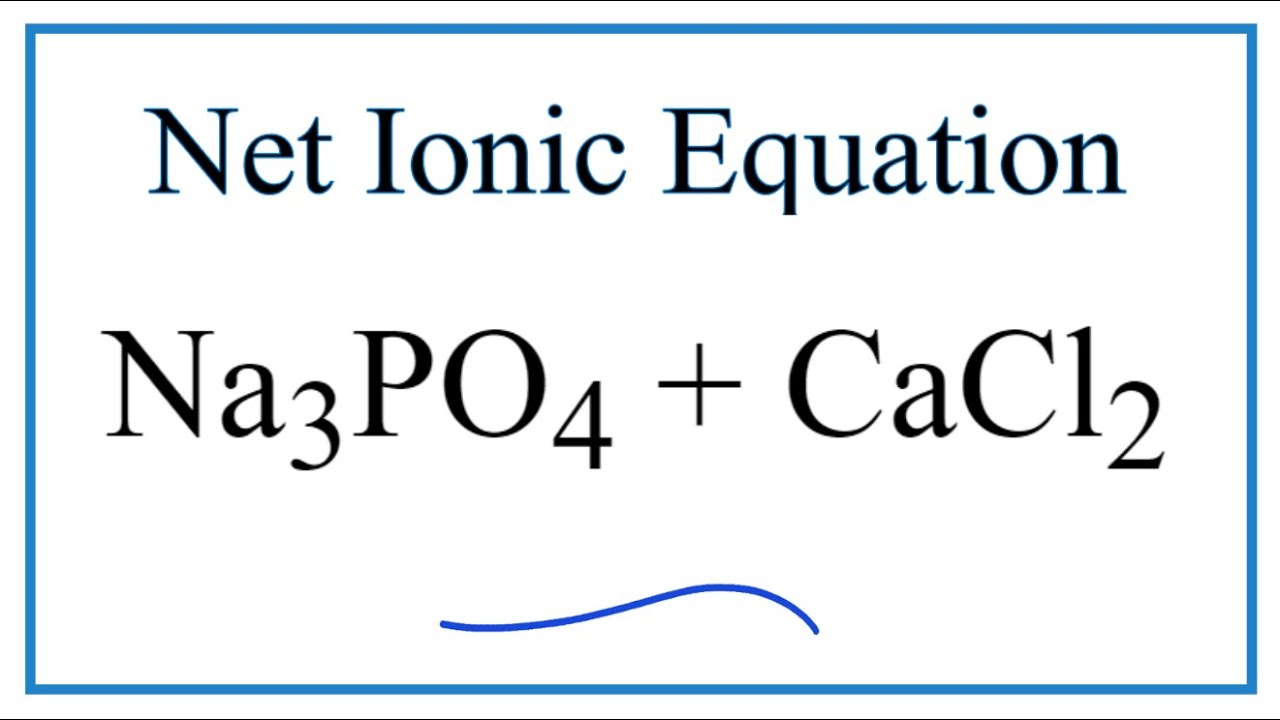OneClass: it has 2 partsh 5. The balanced net ionic equation for the reaction of aqueous calcium chlo...

CaCl2+Na3PO4=Ca3(PO4)2 + NaCl Balanced Equation||Calcium chloride+Sodium phosphate Balanced Equation - YouTube

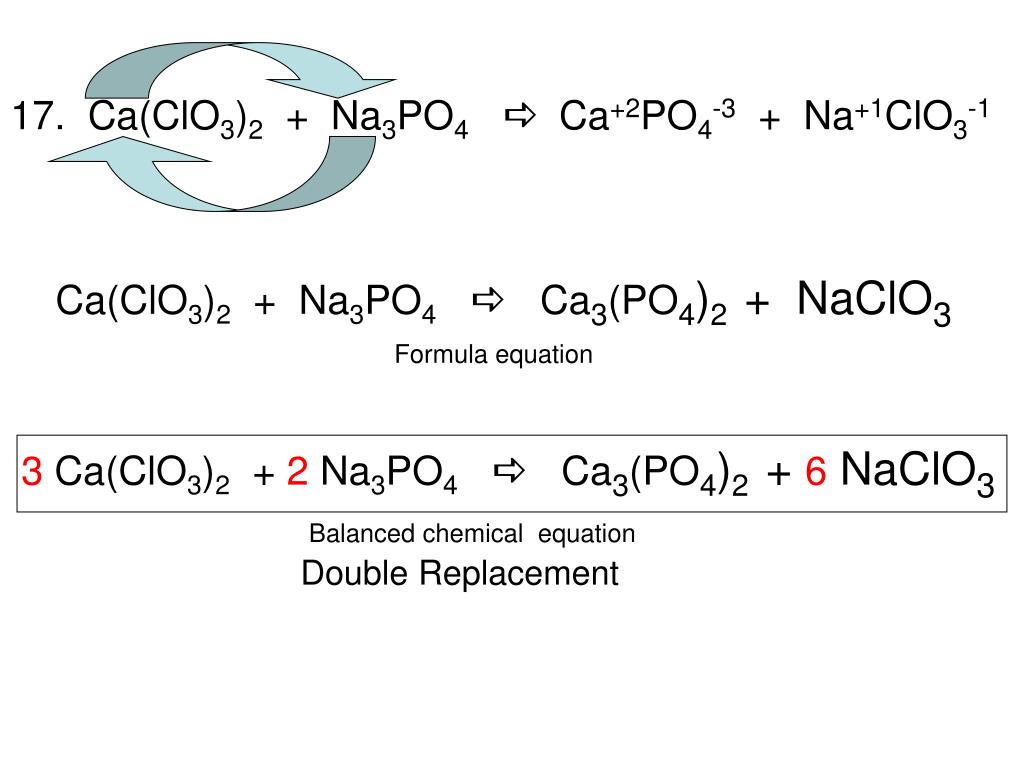
OneClass: I have started this problem but now I am stuck. Please show all work on how the answer is ...

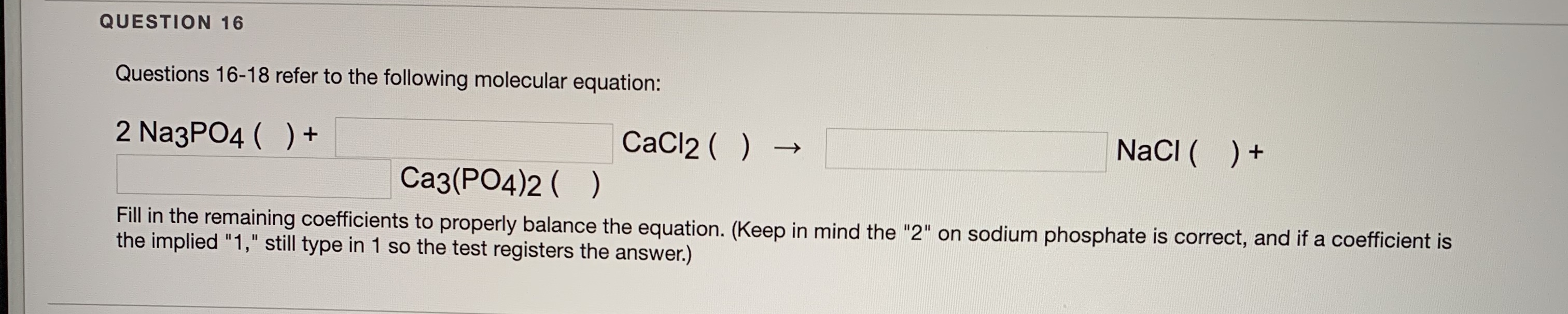
SOLVED: Calcium chloride, CaCl2, and sodium phosphate, Na3PO4 react as follows. 3 CaCl2(aq) + 2 Na3PO4(aq) ⟶ Ca3(PO4)2(s) + 6 NaCl(aq) Molar Masses CaCl2 Na3PO4 Ca3(PO4)2 NaCl (in g mol−1) 110.98 141.96
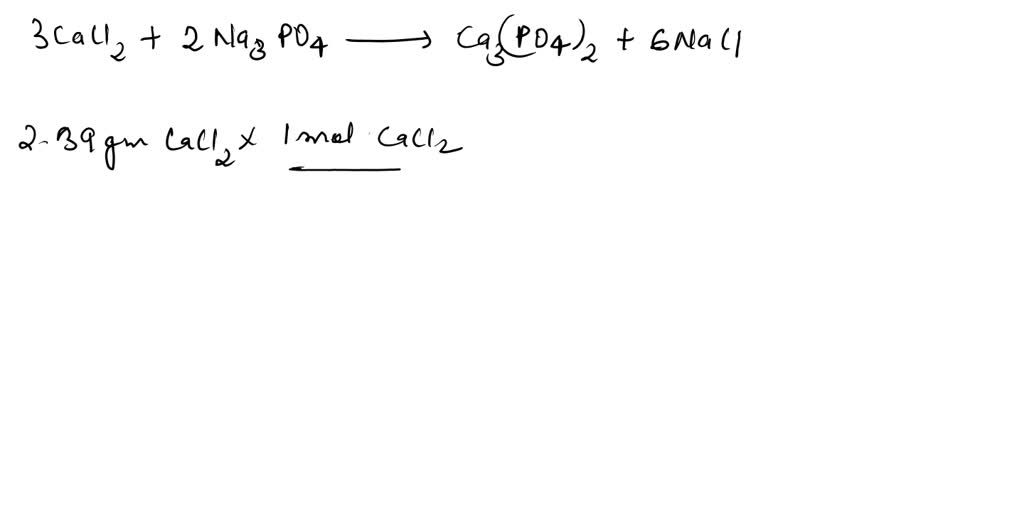
SOLVED: 3 CaCl2(aq) + 2 Na3PO4(aq) -> Ca3(PO4)2(s) + 6 NaCl(aq) 1,50 M 250,0 mL CaCl2 solution is reacted with excess Na3PO4 solution. Mass of the solid product is 36,7 g. What